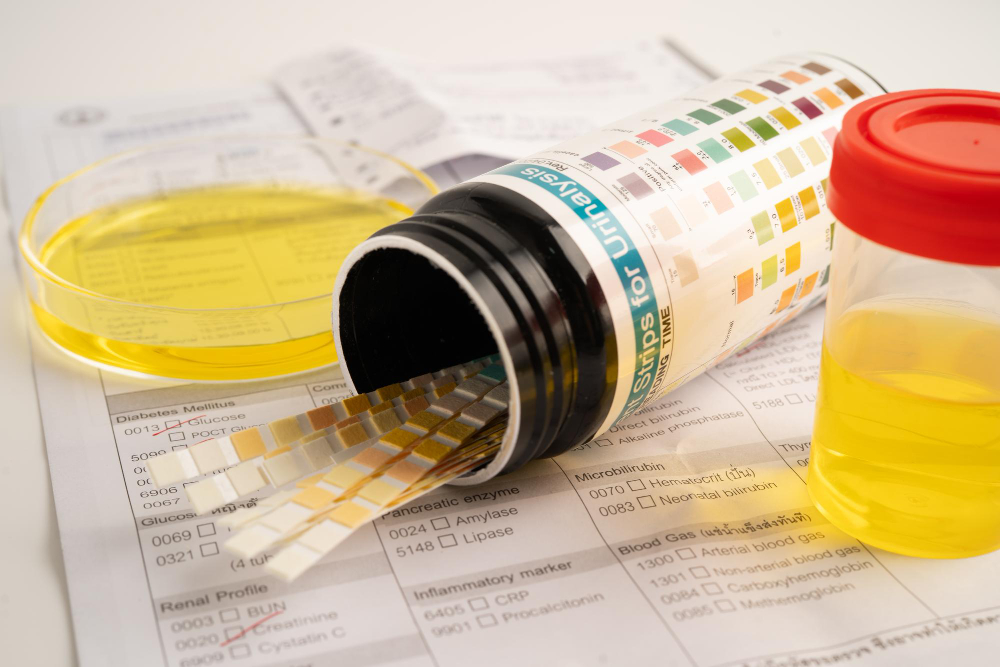
Urinalysis remains one of the most widely used diagnostic tools in laboratory medicine. By examining the chemical and microscopic components of urine, clinicians gain insight into renal filtration, metabolic processes, and urinary tract health. Over time, laboratory technology has expanded from simple reagent strip testing to advanced automated systems capable of measuring urinary biomarkers with high precision.
Two primary approaches dominate modern urine testing: quantitative vs. semi-quantitative urinalysis. Both methods offer valuable information, yet they differ significantly in analytical methodology, operational workflow, and interpretive depth.
Understanding these differences allows laboratories and clinicians to select the most appropriate approach for screening, monitoring, and long-term disease management.
AutoUA® offers the only FDA-cleared quantitative urinalysis system designed to replace dipstick testing, helping laboratories transition from semi-quantitative screening to precise biomarker measurement.
The Historical Role of Semi-quantitative Urinalysis
Semi-quantitative urinalysis developed as a rapid screening method in clinical practice. Reagent dipsticks contain chemically treated pads that react with substances present in urine. When exposed to a sample, these pads change color in proportion to the concentration of specific analytes.
A color scale then converts the reaction intensity into categorical results. Instead of reporting exact concentrations, results are typically expressed as:
- Negative
- Trace
- 1+
- 2+
- 3+
These categories represent approximate ranges rather than exact values. For many decades, this approach served as the primary form of urinalysis because it offered speed, simplicity, and minimal equipment requirements.
Dipstick testing remains common in primary care offices, urgent care settings, and laboratories where rapid screening is needed.
Analytical Foundations of Semi-Quantitative Testing
Dipstick chemistry relies on reagent pads that produce visible color changes when exposed to specific analytes. Each pad contains chemical compounds designed to react with substances such as protein, glucose, blood, or ketones.
The intensity of the color change reflects the approximate concentration within the sample. Automated strip readers can assist interpretation by measuring color intensity digitally, though the result still falls within predefined categorical ranges.
Several analytical limitations arise from this approach. Reagent pad reactions depend on factors such as urine concentration, pH levels, and chemical interference. High vitamin C intake, for instance, may influence certain reactions. Similarly, strongly colored urine may affect visual interpretation.
Because semi-quantitative methods group multiple concentrations into broad categories, small variations in analyte levels often remain hidden within the same classification.
The Analytical Framework of Quantitative Urinalysis
Quantitative urinalysis applies analytical instrumentation to measure urinary biomarkers precisely. Instead of estimating concentration ranges through color reactions, automated analyzers determine exact values using calibrated chemical reactions and optical detection systems.
Measurements are typically reported in standardized units such as milligrams per liter or milligrams per deciliter. This allows clinicians to observe subtle changes in biomarker concentration that semi-quantitative methods cannot distinguish.
Common analytes measured quantitatively include:
- Albumin
- Total protein
- Creatinine
- Albumin-to-creatinine ratios
Because these values represent precise measurements, clinicians can monitor biomarker patterns across multiple visits with far greater resolution.
Sensitivity and Detection Thresholds
One of the most notable differences between the two approaches lies in analytical sensitivity. Semi-quantitative dipsticks detect analytes once concentrations exceed the reaction threshold of the reagent pad.
Values below that threshold may not trigger a detectable color change. As a result, early physiologic changes may remain undetected.
Quantitative analyzers measure analytes across a continuous range of concentrations. This expanded sensitivity allows laboratories to identify low-level biomarker presence that would otherwise appear negative in dipstick testing.
For example, low-level albumin excretion associated with early renal stress may fall below the detection range of reagent strips. Quantitative measurement captures these small elevations and enables clinicians to observe gradual shifts over time.
Precision and Reproducibility
Semi-quantitative categories introduce inherent variability because they represent broad concentration ranges. Two samples within the same category may contain significantly different analyte levels.
For example, two patients might both receive a 1+ protein reading, yet the actual protein concentration in one sample may be substantially higher than the other. The dipstick result cannot distinguish between these differences.
Quantitative measurement eliminates this ambiguity by reporting exact concentrations. Automated analyzers use standardized calibration procedures and controlled chemical reactions to generate reproducible numeric values. This precision improves the ability to monitor longitudinal changes and compare results across laboratory visits.
Operational Workflow Considerations
Operational workflow differs considerably between semi-quantitative and quantitative urinalysis systems.
Dipstick testing requires minimal equipment and can be performed rapidly with limited training. The process typically involves immersing the strip in urine, waiting for color development, and interpreting the result visually or with a strip reader.
This simplicity makes dipsticks useful for high-volume screening environments.
Quantitative urinalysis requires automated instrumentation capable of processing samples through chemical analysis. While this equipment represents a greater technological investment, modern analyzers integrate smoothly with laboratory workflows.
Automated urinalysis systems can process multiple samples simultaneously and transmit results directly to laboratory information platforms. This digital integration simplifies result reporting and long-term data storage.
Clinical Interpretation and Diagnostic Depth
Semi-quantitative testing functions primarily as a screening method. Its categorical results indicate the presence of certain substances but provide limited information about concentration changes over time.
Quantitative urinalysis offers a deeper level of diagnostic detail. Exact concentrations allow clinicians to detect subtle biomarker shifts that may signal evolving physiologic changes.
This level of detail becomes particularly valuable in chronic disease monitoring. For instance, patients with diabetes or hypertension often undergo periodic urine testing to evaluate kidney health.
Precise measurement allows clinicians to track gradual changes in albumin excretion or protein concentration. These patterns may reveal early renal stress long before symptoms appear.
Longitudinal Monitoring and Trend Analysis
One of the strongest advantages of quantitative measurement lies in its ability to support trend analysis. Numeric data can be compared across multiple visits, revealing patterns that categorical dipstick results may conceal.
Examples of trends detectable through quantitative testing include:
- Gradual increases in albumin-to-creatinine ratios
- Persistent low-level protein excretion
- Reduction in urinary protein following therapeutic intervention
Because semi-quantitative categories compress multiple values into broad ranges, these subtle changes often remain hidden.
Trend analysis plays a central role in preventive medicine and chronic disease management. Quantitative data provides the resolution required for meaningful interpretation.
Move Beyond Dipsticks with Quantitative Urinalysis
Traditional dipstick testing offers quick screening, but modern laboratories often require more precise data. AutoUA® is the only FDA-cleared quantitative urinalysis system designed to replace semi-quantitative dipsticks with objective numeric measurement. The platform analyzes urine chemistry and particle elements while generating standardized ratios such as albumin-to-creatinine through an automated workflow.
AutoUA® is compatible with many leading clinical chemistry analyzers, allowing laboratories to integrate quantitative testing without major workflow disruption. Direct LIS connectivity also allows clinicians to review historical values and identify trends more easily.
Contact our team to learn more about AutoUA®, request additional information, or schedule a product demonstration.